
Molecular genetic testing approaches can include a combination of gene-targeted testing (multigene panel) and comprehensive genomic testing (exome sequencing, genome sequencing). (2) Identification of a hemizygous IL2RG variant of uncertain significance does not establish or rule out the diagnosis. Reference to "pathogenic variants" in this GeneReview is understood to include likely pathogenic variants. Note: (1) Per ACMG/AMP variant interpretation guidelines, the terms "pathogenic variant" and "likely pathogenic variant" are synonymous in a clinical setting, meaning that both are considered diagnostic and can be used for clinical decision making. The diagnosis of X-SCID is established in a male proband with suggestive findings and a hemizygous pathogenic (or likely pathogenic) variant in IL2RG identified by molecular genetic testing (see Table 2). Pending establishment of the diagnosis and initiation of treatment, parents and other care providers for all infants need to avoid all of the following: For other genetic causes of an abnormal NBS possibly indicating SCID, see Differential Diagnosis.Įnsuring the safety of the infant. 
Note: This chapter specifically focuses on X-linked severe combined immunodeficiency (X-SCID), one genetic cause of SCID. Newborns with an abnormal NBS require immediate subspecialty immunology evaluation at a center with expertise in the diagnosis of SCID and its genetic causes, and in SCID treatment protocols, including hematopoietic stem cell transplantation (HSCT) or gene therapy. 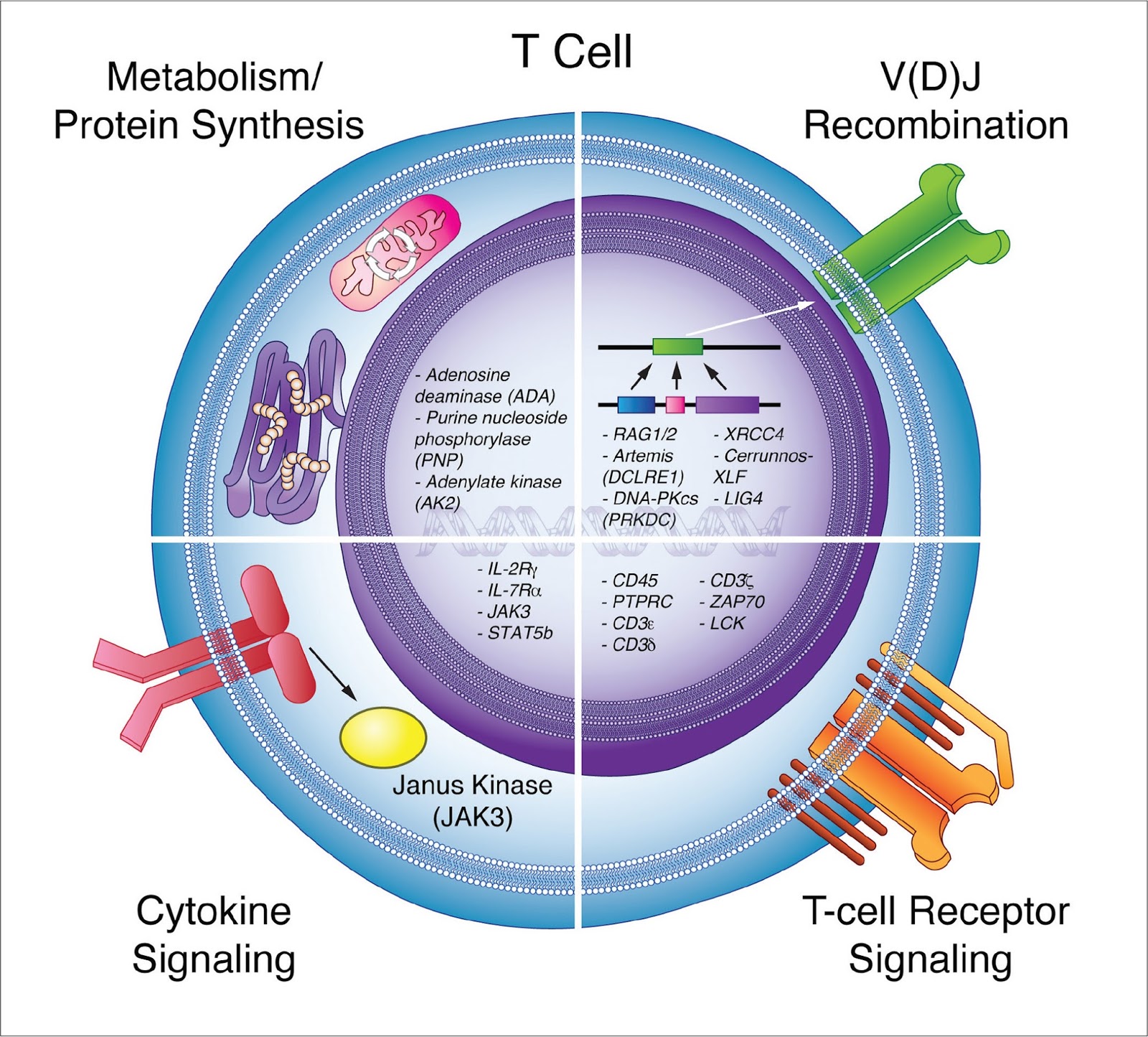
In the US, the newborn screening agency in each state determines the TREC threshold that indicates possible SCID. An abnormal NBS result (low/absent TRECs) indicates clinically significant autologous T lymphocytopenia (<1500 T cells/μL). SCID newborn screening uses a blood spot to measure T-cell receptor excision circles (TRECs) to detect T-cell lymphopenia. Scenario 1: Abnormal SCID Newborn Screening (NBS) – Typical X-SCIDĪs of December 10, 2018, all newborns in the US, including all 50 states, the District of Columbia, and the Navajo Nation, are screened for a group of conditions characterized by severe combined immunodeficiency (SCID), (adapted from the PIDTC criteria ). If prenatal testing has not been performed, an at-risk newborn male should immediately be placed in a safe environment and tested for the familial IL2RG pathogenic variant to allow earliest possible diagnosis and treatment. If HSCT involved conditioning chemotherapy, long-term monitoring of vital organ function and neurodevelopmental progress is also warranted.Īgents/circumstances to avoid: To ensure the safety of affected individuals of all ages pending definitive treatment to achieve immunocompetence, parents and other care providers need to assure that the following are avoided: breast-feeding and breast milk (pending clarification of maternal CMV status) exposure to young children, sick persons, or individuals with cold sores crowded enclosed spaces live viral vaccines for the affected individual as well as household contacts transfusion of non-irradiated blood products areas of construction or soil manipulation.Įvaluation of relatives at risk: When the IL2RG pathogenic variant causing X-SCID in the family is known, prenatal testing of at-risk male fetuses may be performed to help prepare for optimal management of an affected infant at birth. 
Surveillance: After successful HSCT, routine monitoring of affected males every six to 12 months regarding lineage-specific donor cell engraftment growth, immune, and lung function and any gastrointestinal and/or dermatologic issues. Treatment depends on the degree of infectious complications and the presence of immune dysregulation and/or autoimmunity, and requires subspecialty immunologic care to assist in the diagnosis and choice of antimicrobial and immune-suppressive therapies.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
